December 2025. Your diagnostic AI algorithm is a technical masterpiece. You have clinical proof of its performance(90% diagnostic success rate). Yet if you don’t act now, it will be illegal after August 2, 2026.
The paradox of AI in healthcare is no longer technology, but regulation. The era of rapid innovation is giving way to the era of Urgent Compliance.
At JuliaShift, we observe that the major risk for scale-ups is not piracy, but regulatory failure due to clinical bias and the impendingEU AI Act.
In this article, we decipher the three essential elements for transforming this risk into resilience: the critical August 2, 2026 deadline, the legal danger of unmanaged data bias, and the requirement for a robust Human-in-the-Loop.
1. The Sanction Clock: AI Act & High-Risk Status
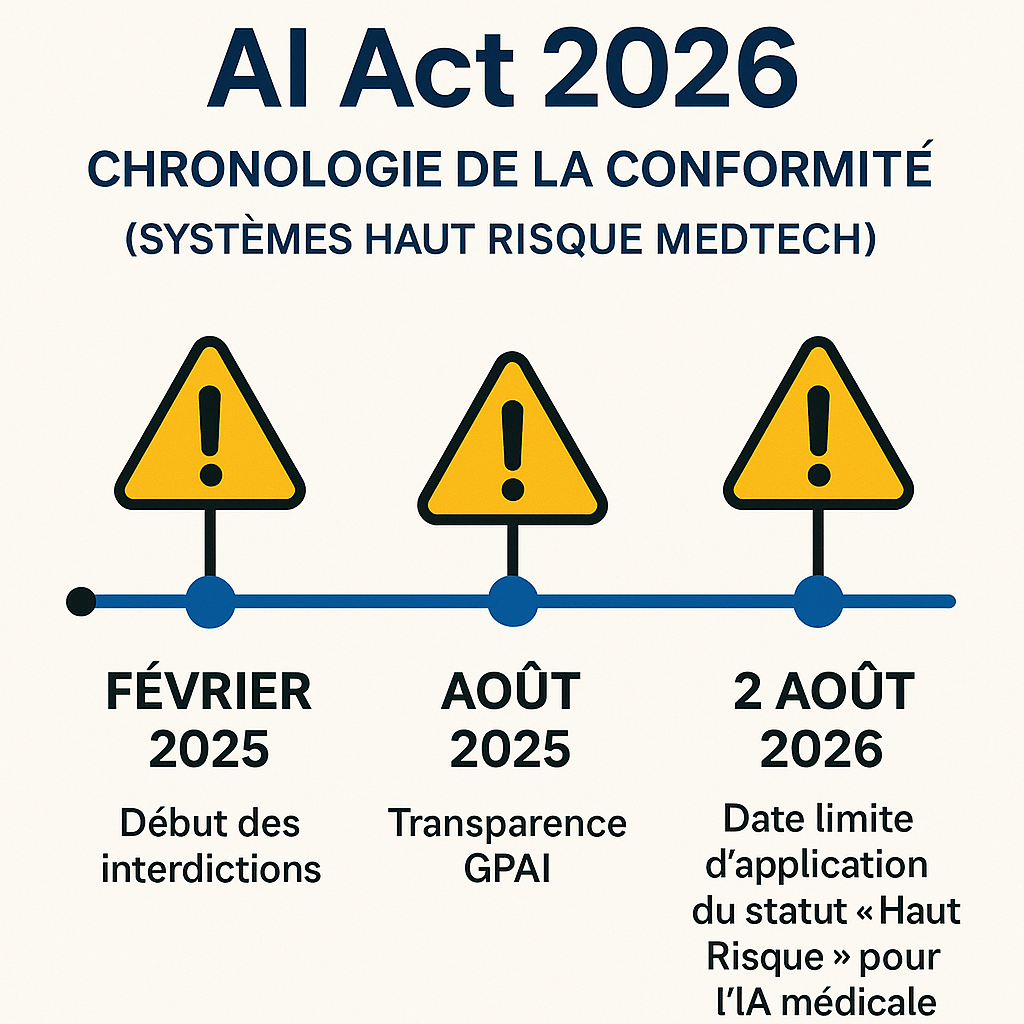
Regulation 2024/1689(AI Act) is not a suggestion, it’s a law that’s gradually coming into force. Forget the wait-and-see strategy.
- The “High Risk” Classification has been implemented: All AI systems for diagnosis and clinical decision-making are classified as HIGH RISK. This requires a continuous Risk Management System (RMS) and full documentation, right from implementation.
- Deadline: Application of the rules for high-risk systems is set for August 2, 2026. This is the ultimate deadline for aligning your engineering processes and technical documentation with European requirements (Source: CERTEAFILES, PMC).
2. The Judicial Danger of Bias: Discriminating AI
The AI Act sanctionsinequity. Your AI could be non-compliant if it underperforms on part of the European population.
- Le Chiffre de l’Inégalité: Lack of representative data (gender or skin color bias) degrades diagnostic performance. The error rate can rise from 1% (on the majority population) to a dangerous 35% on under-represented populations. This represents a risk of clinical invalidation and legal sanction.
- Hallucination: generative AI systems (LLMs) have high diagnostic performance, but produce probabilistic content. The risk ofhallucination (invented or obsolete data) requires strict supervision to avoid clinical error.

3. Human Control: The Resilience Imperative
Target Keywords : Human-in-the-Loop Légal, Gouvernance IA MedTech, Responsabilité médicale IA.
The AI Act requires supervision. The HAS (Haute Autorité de Santé) provides the practical framework for this obligation in the French context.
- HIL is the Law: The AI Act makes “Human-in-the-Loop” (HIL ) legally mandatory for all critical diagnoses. The physician retains final responsibility (Source: Ariis Brief).
- The HAS framework: The HAS recommends that clinicians appropriate these tools by following the 4 AVEC axes (Apprendre-Vérifier-Estimer-Communiquer) to guarantee proper use (Source: Guide HAS IA générative Oct 2025). This is the governance framework you need to integrate.
Strategic conclusion: Anticipating risk
Now is not the time for technological justification, but for regulatory and ethical security. Your most profitable investment is inauditing your data sets and your chain of compliance.
Open question to MedTech CEOs: Who validated the absence of bias on your training set? Is your CIO ready for the AI Act audit in 2026?
🎯 CALL TO ACTION
Are you structuring the compliance of your AI systems for 2026?
We support you in theClinical and Regulatory Robustness Audit of your existing AI systems. Don’t let a clinical bias derail your market strategy.
👉 Book your strategy consultation
Would you like to receive L’Éclaireur e-Santé every month? Read all our analyses in our newsletter.
🎯 Going further
Are you structuring a MedTech fundraiser?
Download our free strategic reports:
- BPI France 50-point compliance checklist
- Timeline 0-6 months pre-emergence
- 3 startup cases (seed → series A)
- Frameworks valorisation multiples Revenue
📥 Download your free reports → Blueprint MedTech
About the author
Nicolas Schneider is a strategic consultant in digital healthcare transformation and founder of JuliaShift. With 17 years’ experience at the Service de Santé des Armées and 8 years in digital transformation consulting, he assists MedTech startups and healthcare establishments in their financing strategy, structuring pharma partnerships and preparing for fund-raising.
Specialties: healthcare innovation financing, MedTech fund-raising structuring, pharma industrial partnerships, IA regulatory compliance.