From idea to market, e-health startups face a complex, regulated sector.
The key figure: 70% of e-health startups fail before the end of year 3. Not for lack of technology. Strategy.
Why? Two classic mistakes: underestimating regulation (RGPD, medical devices, ANSM) and rushing development without validating the real need. The result? Budget squandered. Exploding time-to-market. Fatal product-market mismatch.
Good news: these pitfalls are avoidable. Thanks to a proven methodology and a rigorous sequence, you can turn your innovation into a viable business.
By the end of this article, you’ll have a clear roadmap: the 7 concrete steps. Not theory. Pragmatism in the field.
Why 70% of e-health startups fail
The healthcare sector is not like traditional tech. Three fundamental differences explain this failure rate.
Difference 1: Regulation kills improvisation
In pure tech, you launch, you iterate, you fix the problems afterwards.
In healthcare, it’s the other way around. Regulation comes first.
In concrete terms: RGPD = ultra-sensitive patient data. Medical device = CE certification required (6-18 months). ANSM = if you’re dealing with diagnostics, opinion required. HDS = data hosted in France (costly).
So, 65% of startups discover these obligations too late. The average extra cost? 40% of the initial budget. That’s catastrophic.
Difference 2: The sales cycle is considerably longer
In classic B2B tech, you sell in 3-6 months.
Healthy ? 12-24 months. Why? Hospital decision-makers test rigorously. They demand safety audits. Physician validation. Ethics committees. Each layer adds time.
Direct implication: You have to finance two years with no revenue. Few startups survive this cash flow test.
Difference 3: Real adoption requires organizational change
A surgeon will only accept a new tool if he understands how it changes his daily workflow.
So you’re not just selling a product. You’re selling organizational change, training and ongoing support.
Consequence: Successful startups invest 30-40% of their resources in adoption, not just technical development.
The three pillars before launching: diagnosis, regulation, strategy
Before you can code a single line, you need to validate three foundations. Without these three pillars, the risk of failure climbs to 80%.
Pillar 1: Rigorous market diagnosis
The fundamental question: Are you trying to solve a real need or are you pursuing a personal intuition?
To respond, conduct 25-30 interviews with target decision-makers (university hospitals, firms, competing startups). Map your direct and indirect competitors. Evaluate willingness-to-pay: what price do decision-makers really accept?
So, after 4-6 weeks, you’ll have a clear understanding of the user journey and a clear differentiation vs. competition. Budget required: €5-10K.
The warning sign: If you find less than 20 people ready to talk, it’s a bad sign. Pivot or give up.
Pillar 2: Early regulatory mapping
The real questions : What regulatory framework does your product depend on? What legal obstacles can you anticipate right now?
Start by identifying whether your product is a medical device (if so → CE certification required). Are you processing patient data (if so → RGPD + HDS mandatory). Do you perform diagnosis or treatment? (If yes → prior ANSM opinion). Do you host data? (If yes → French servers required).
So, in 2-3 weeks, build a compliance roadmap with clear milestones. Mobilize a specialized healthcare lawyer (3-5K€). That way, you’ll avoid costly surprises later on.
The signal: You need to be able to answer these regulatory questions before raising funds. It’s a deal-killer for investors.
Pillar 3: Clear positioning strategy
The strategic question: Who exactly is your ideal customer? What specific problem are you solving for them?
Build a positioning matrix. Identify your type of decision-maker (hospital, practice, start-up, industry). Identify who allocates the budget (HR, IT, clinical management, R&D). Target the specific problem you’re solving (operational efficiency, regulatory compliance, patient satisfaction). Clearly measure the ROI benefit (reduced time, lower costs, higher revenues).
Result: memorable positioning in a single sentence. For example: “AI breast cancer diagnosis for radiologists” (precise) vs. “digital health platform” (too vague).
In 3-4 weeks, you should have this positioning crystallized. The simple test: does a non-tech decision-maker understand your solution in 30 seconds?
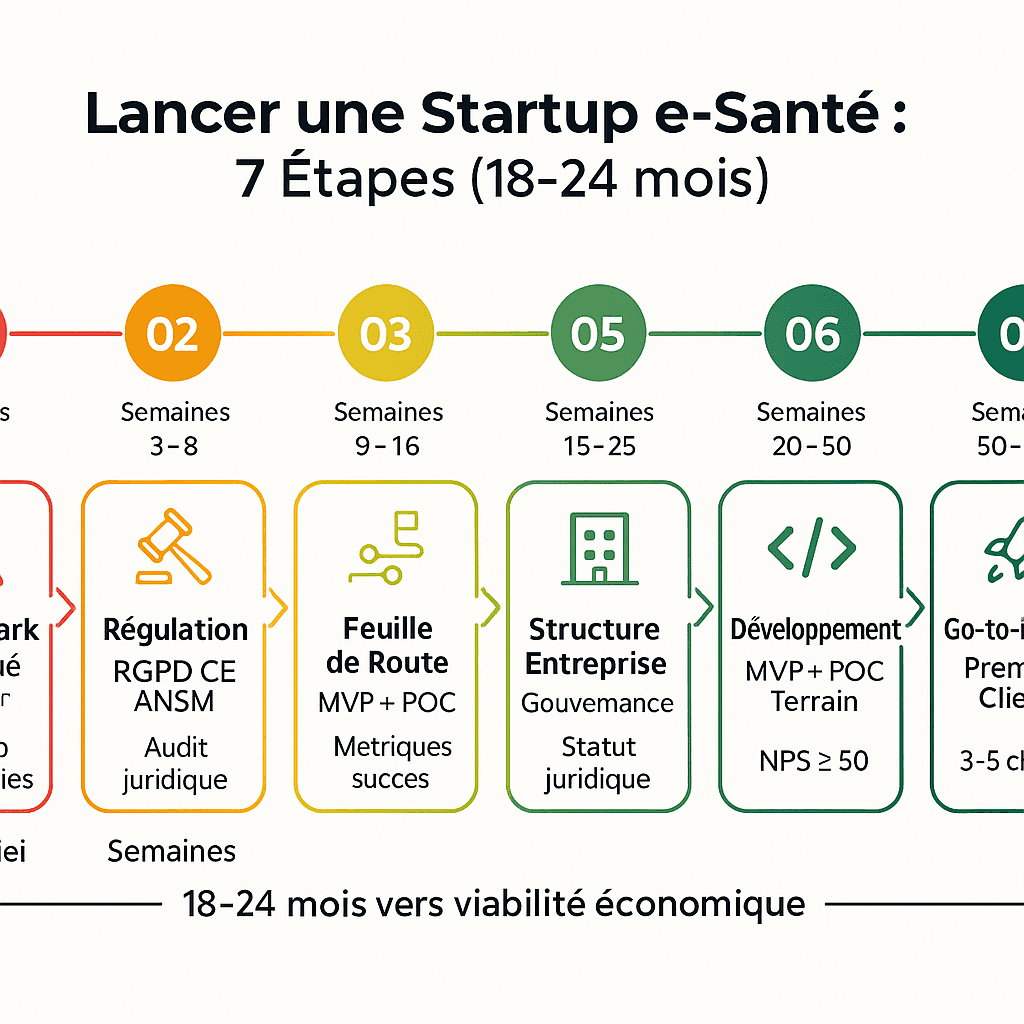
7 concrete steps: from idea to market
Now for the roadmap itself. Each step builds on the previous one.
Stage 1: Structured benchmark + market validation (Weeks 1-6)
The goal: To confirm that the need is real, not just your perception.
Conduct 25-30 interviews with target decision-makers to validate their problem. Analyze 10-15 direct and indirect competitors to verify your differentiation. Map the detailed user journey. Measure willingness-to-pay by asking decision-makers to rate you on a scale of 1-10.
Success criteria: At least 70% of decision-makers identify the problem you’re targeting. Your solution is clearly differentiated vs. competition. The willingness-to-pay score reaches ≥ 7/10.
If not validated : You can pivot or give up. Better to find out early.
Stage 2: Regulatory mapping + compliance plan (Weeks 3-8)
The aim: to turn regulatory obstacles into a competitive advantage, not a barrier.
Have your product audited by an e-health lawyer. Define its classification (is it a medical device?). Build a certification roadmap (CE, HDS, RGPD audit). Identify regulatory “early wins”: easy points to validate quickly to gain the trust of your partners.
Success criteria: Product classification is unambiguously confirmed. Regulatory roadmap with clear, realistic milestones. The compliance budget is defined and costed. No costly surprises.
Budget: 8-12 weeks in total.
Stage 3: Product roadmap + structured POC (Weeks 9-16)
The aim: to validate technical feasibility AND clinical fit in a real-life environment.
Define your MVP (essential features only, nothing extra). Plan a short PoC: 3-4 months in a real hospital or practice, not in a lab. Establish measurable success metrics. They should cover three dimensions: clinical (medical usefulness), UX (ease of use), technical (system robustness). Estimate the budget for phase 1, including development and testing.
Success criteria: Your MVP is defined and non-negotiable. A PoC in CHU/cabinet is confirmed in writing. Metrics are precise and ambitious (e.g. NPS ≥ 50, adoption ≥ 80%).
Duration: 8-10 weeks.
Stage 4: Structuring hybrid financing (Weeks 10-20)
Objective: Secure cash flow for 18-24 months. It’s critical.
Map out the sources of funding best suited to the healthcare sector. Start with public funding: BPI France Santé Numérique (subsidies), Fonds Patient Autonome (medico-social), AMI e-Parcours (patient pathways). Then target private investors: healthcare business angels, specialized VC funds (Demeter, Sofina, etc.). At the same time, negotiate institutional CHU/clinic partnerships to generate early deals.
Concrete actions: Structure a complete BPI file (executive summary, roadmap, detailed budget). Test your pitch with 5+ investors. Sign ≥ 1 institutional partnership.
The result: This approach increases your chances of financing by 63%.
Stage 5: Structuring the company + governance (Weeks 15-25)
The aim: to lay solid legal and organizational foundations.
Choose the right legal status (SARL, SAS, why this or that form?). Structure your governance: who sits on the board? Do you need to set up a health ethics committee? Prepare your key recruitments (CTO, regulatory manager, first sales representative). If you’re developing a medical device, set up quality processes right away.
Success criteria: Legal status is chosen and registered. Roles and responsibilities are clear (who decides what?). Health quality processes are documented.
Duration: 8-10 weeks.
Stage 6: Product development + field testing (Weeks 20-50)
The aim: to create a functional MVP, tested with real users in real-life conditions.
Develop your MVP (typically 4-6 months). At the same time, test RGPD and HDS (data security) compliance. Launch PoC in hospital or practice: 50-100 users, 3-4 months. Collect structured feedback. Iterate rapidly: every week of adjustments counts.
Key success criteria: MVP meets RGPD/HDS requirements without compromise. At least 80% of PoC users say “it’s useful”. Your NPS (Net Promoter Score) reaches ≥ 50. Zero major security incidents during the test.
Duration: approximately 30 weeks.
Stage 7: Go-to-market + progressive scaling (Weeks 50-80+)
The goal: Move from successful PoC to first full-scale customers. This is the moment when you validate your business model.
Define your go-to-market strategy: are you going to sell to hospitals, practices or directly to patients? Deploy your first real customers. Set up contracts, support and training. Recruit a sales manager if necessary. Adapt your business model according to actual customer feedback. Anticipate European expansion: regulations differ from country to country.
Success criteria: You have signed 3-5 confirmed customers. Your monthly revenue is at least €10K. Your churn rate is below 5% (which means your customers are staying). Your 12-month product roadmap is planned and validated.
Duration: 30+ weeks (continuous growth).
Synthetic timeline: from idea to viability
Semaines 1-6 : Diagnostic + validation marché
Semaines 3-8 : Cartographie réglementaire
Semaines 9-16 : Roadmap produit + PoC structurée
Semaines 10-20 : Financement (BPI, angels, VC)
Semaines 15-25 : Structuration entreprise
Semaines 20-50 : Développement MVP + PoC terrain
Semaines 50-80+ : Go-to-market + premiers clients
Total : 18-24 mois avant viabilité économique réelle
Key resources to know
To structure your journey, identify the official aids :
Financing and subsidies :
- BPI France Santé Numérique (e-health subsidies)
- Fonds Patient Autonome (medico-social)
- AMI e-Parcours (patient pathway)
Regulation and compliance :
Innovation ecosystem :
- Health Competitiveness Clusters (regional clusters)
- e-health incubators (local networks)
Our approach: three key levers
At JuliaShift, we guide e-health startups through these stages.
Lever 1: Market diagnosis + POC validation
Confirm the need before investing in development.
We conduct structured interviews, analyze competition, validate willingness-to-pay.
Lever 2: Turn regulation into an advantage
Anticipate regulatory obstacles and turn them into a commercial advantage.
We structure compliance roadmaps, prepare certification dossiers and guide ANSM/HAS interactions.
Lever 3: Hybrid financing + go-to-market
Access to public funding + private investors + first customers.
We structure BPI files, facilitate investor pitches, negotiate CHU partnerships.
Launching an e-health startup?
Are you looking to validate your idea before making a major investment?
Do you have an MVP but are struggling to raise funds or find your first customers?
We structure: market diagnosis + regulatory roadmap + financing strategy + network access.
30-minute consultation to diagnose your situation.
🎯 Going further
Are you structuring a MedTech fundraiser?
Download our free strategic reports:
- ✓ BPI France 50-point compliance checklist
- ✓ Timeline 0-6 months pre-emergence
- ✓ 3 startup cases (seed → series A)
- ✓ Frameworks valorisation multiples Revenue
📥 Download your free reports → Blueprint MedTec
Take action and secure your international expansion!
Your MedTech Series A/B can become a European leader. JuliaShift guides you with its Diagnosis – Execution – Control protocol to transform strategy, financing and clinical adoption into concrete results.
🔦 Discover the MedTech Blueprint → Strategic keys to your international deployments
🧩 Explore our Portfolio → Real projects, validated revenues, trained teams
🛎️ Book your 4D Diagnostic → Move to execution with a clear, prioritized plan
About the author
Nicolas Schneider is a strategic advisor in digital healthcare transformation and founder of JuliaShift. With 17 years’ experience at the French Army Health Service and 8 years in digital transformation consulting, he assists MedTech startups and healthcare establishments in their financing strategy, structuring pharma partnerships and preparing for fund-raising.
Specialties: healthcare innovation financing, MedTech fund-raising structuring, pharma industrial partnerships, IA regulatory compliance.