Europe’s medical device certification strategy: why 76% of startups blow their budgets
Sophia Genetics spent an extra 250,000 euros and lost 12 months migrating from IVDD to IVDR in 2024. For a MedTech startup with a limited runway, it’s the difference between scale and shutdown. The Nasdaq-listed company had underestimated the regulatory transition. As a result, its competitors positioned themselves in the meantime.
The European Union’s 2025 figures reveal an even more worrying reality. Out of 28,489 certification applications registered, only 12,177 certificates were issued. This represents a gap of 57%. Even more alarming: 58% of total delays come from the manufacturers themselves. The cause? Incomplete documentation. In other words, the problem is not just regulatory.
Structuring an optimized medical device certification strategy can reduce your costs by 30 to 40%. Your turnaround time can be reduced by 4 to 6 months. This guide decodes the 6 fatal errors observed in 501 European MedTech startups. It provides an actionable framework for optimizing your regulatory roadmap without blowing your budget.
Let’s start by understanding why the choice of entrance market determines 70% of your certification budget.
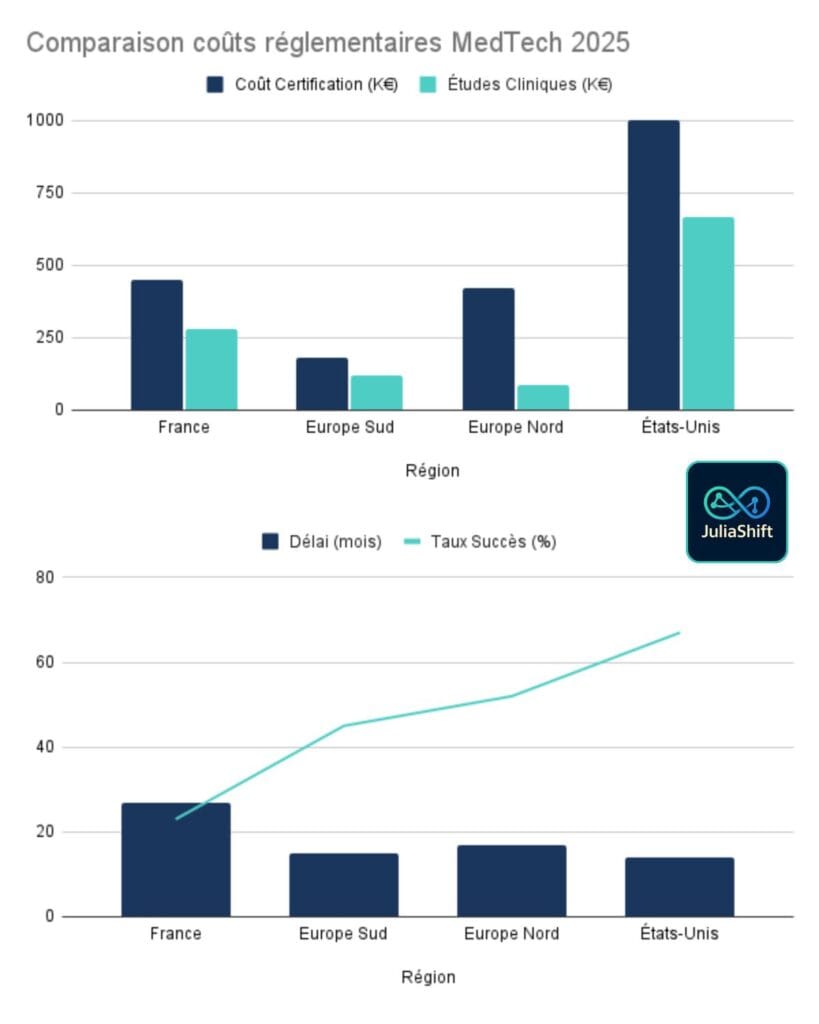
The European paradox – why the entry market determines your budget
European regulations allow any member country to obtain CE marking. In theory, this should standardize costs. However, certification in France costs 450,000 euros. In Italy, the same Class IIa device requires 180,000 euros. This disparity represents a difference of 270,000 euros. For a pre-seed startup, that’s an extra 18 months’ runway.
Three factors explain this structural gap
Firstly, the capacity of notified bodies varies considerably. France has 3 notified bodies, which are often saturated. Germany has 8 with greater capacity. Italy offers 4 bodies with shorter queues.
Secondly, industry experience plays a critical role. German organizations process 2.3 times more Class IIb devices per year. This difference translates into faster expertise. Examinations run more smoothly.
Thirdly, lead times differ radically. Lead time is the time before the organization initiates the examination of your file. BSI UK/Europe estimates 18 to 24 months. TÜV Italy suggests 10 to 12 months.
EU 2025 data confirm this reality
The actual average time required to obtain Class IIa or IIb certification is between 13 and 18 months. The hourly cost of expertise varies between 250 and 430 euros per hour. The success rate at first submission reveals a major disparity: 23% in France versus 52% in Northern Europe.
This last figure means that 77% of French startups have to rework their applications. This generates substantial additional costs. When you miss the first submission, you lose 3 to 6 months. You spend an extra 50,000 to 75,000 euros.
Sophia Genetics illustrates these challenges
Sophia Genetics (Nasdaq: SOPH) obtained its IVDR certification in August 2024. The company had to recertify its legacy IVDD device. This regulatory transition generated an additional cost of 250,000 euros. The delay? An additional 12 months.
During this period, several competitors positioned themselves in key market segments. Sophia’s first-mover advantage was eroded. German university hospitals signed up with other suppliers. The commercial delay was irreversible.
Does your certification strategy include the selection of a strategic notified body? Or does it remain focused solely on your home market? This question often determines the difference between a successful lift and a premature shutdown.
The 6 mistakes that blow up your certification budget
Analysis of 501 European MedTech startups reveals 6 recurring errors. These errors add up to between 150,000 and 350,000 euros in hidden costs. That’s the equivalent of 12 to 24 months of wasted runway. Let’s take a closer look at each of them, with documented case studies.
Error #1: Incorrect device classification
Impact: +150,000 euros | +6 months | 45% of startups affected
A French diagnostic start-up has self-classified its IVD as Class I. It thought it could benefit from self-declaration without a notified body. During the pre-certification audit, the notified body reclassified the product as Class IIb. Clinical data were insufficient to confirm the diagnostic claims.
The cascading impact was brutal. The start-up had to conduct ad-hoc clinical studies. Investment: 80,000 to 150,000 euros. The additional delay reached 6 months. Meanwhile, competitors were expanding into the German market.
The solution was avoidable. A pre-submission meeting with a notified body costs 5,000 to 8,000 euros. It would have confirmed the correct classification BEFORE any development investment. The savings: 40% of the total time and 150,000 euros.
Error #2: Insufficient clinical evidence
Impact: +200,000 euros | +8 months | 62% affected
Founders often think that a literature review will suffice. This is true for Class I, sometimes for Class IIa. For Class IIb, the regulatory reality is different. These devices require their own patient data. Typically, you need to include 60+ participants. The protocol must be validated by an IRB ethics committee.
The EU Survey 2024 reveals that 60 out of 501 companies have had their certification suspended. The reason? Insufficient evidence. This suspension often occurs at an advanced stage. It forces the company to conduct urgent clinical studies. Worse still, it compromises credibility with investors who were expecting an imminent launch.
Error #3: Wrong choice of notified body
Impact: +100,000 euros | +4 months | 38% of startups
One startup submitted its file to an organization with an 18-month lead time. Its strategic competitor chose a German agency with a 13-month lead time. As a result, the competitor benefited from a 5-month lead on the market. He captured the first-mover advantage. The first hospital contracts had already been signed.
Data for 2025 show that, out of 51 notified bodies designated for MDR, capacity remains heterogeneous. BSI UK/Europe has a lead time of 18 to 24 months. TÜV SÜD Germany suggests 13 to 15 months. Some Italian bodies maintain 10 to 12 months.
The lesson is clear. Don’t choose your notified body by default. Analyze capacity strategically. Ask for a written capacity statement. Compare 3 to 4 organizations before deciding.
Error #4: Incomplete documentation
Impact: +75,000 euros | +3 months | 58% of total lead time
This is the most frequent error. It accounts for 58% of total delays, according to EU 2025 data. The statistic is alarming: more than half the delays come from the manufacturers themselves. The notified body is not to blame. The reason? Incomplete submissions requiring multiple round trips.
Each overhaul cycle costs 15,000 to 25,000 euros. It adds 4 to 6 weeks to the calendar. After 3 cycles, you’ve lost 3 months and 75,000 euros.
The solution exists. Carry out a documentation checkpoint 2 weeks before official submission. The notified body returns informal comments. You correct BEFORE formal rejection. You save several costly cycles.
Error #5: Regulatory transition ignored
Impact: +250,000 euros | +12 months | 35% affected
Sophia Genetics is a perfect example of this pitfall. Maintaining a legacy IVDD device while having to recertify it under IVDR amounts to a double layer of costs. You pay for the old certification. You also pay for the new recertification.
The critical MDR deadline is December 31, 2027. It concerns Class III devices and Class IIb implants. Startups that wait too long risk commercial paralysis. They can no longer sell their legacy devices. They don’t yet have their new certification.
Anticipate these transitions. Build them into your roadmap from the outset. Budget for recertification. Don’t get caught out 18 months before the deadline.
Error #6: ISO 13485 non-compliant quality system
Impact : +120 000 euros | +5 months | 48% of startups
The fatal mistake is to develop the product on the assumption that “we’ll deal with the QMS later”. This approach generates an additional cost of 120,000 euros. It also adds 5 months of delay.
Reality is implacable. Design controls, traceability and ISO 14971 risk management are prerequisites. They are not administrative formalities. Notified bodies detect major non-conformities. They suspend certification until they have been fully corrected.
This suspension lasts 5 months or more. During this time, the company cannot sell, raise funds or make commercial progress. Cash burns up with no revenue generation.
How many of these 6 mistakes do you recognize in your current roadmap? Honesty about these vulnerabilities is the first step towards an optimized strategy.
To find out more about the challenges of overall structuring, read our article on how to structure your e-health project along 6 pillars to avoid failure.
Decision-making framework – which class, which entry market?
The classification of your medical device determines your entire strategy. Costs, lead times, clinical requirements, optimal market: everything stems from this classification. And yet, it often remains unclear until confrontation with the notified body.
Class IIa: moderate to slight risk
These systems cost between €200,000 and €300,000 to certify. The average lead time is 13 to 18 months. Clinical evidence is based on a literature review supplemented by moderate testing. The success rate for the first submission is 72%.
France and Germany remain the optimum entry markets. Notified bodies are very familiar with these markets. Examples of Class IIa devices: monitoring devices, diagnostic software, certain medical wearables.
Class IIb: moderate to high risk
The cost is between 300,000 and 500,000 euros. Lead times range from 15 to 20 months. At this stage, proprietary clinical data becomes mandatory. You need to include patients in validated protocols.
The success rate drops to 58%. This reflects a much higher regulatory requirement. For Class IIb, Germany is the optimal priority market. Why? Its 8 notified bodies are experienced in this risk class.
A relevant sequential strategy: target Germany at time t+18 months. Then attack France at t+30 months. You maximize market access. You optimize costs.
Examples of Class IIb devices: implantable sensors, surgical tools, certain interventional radiology devices.
Class III: high risk
The cost often exceeds 500,000 euros. It can reach 1 million euros. Lead times range from 20 to 36 months. Full clinical trials are becoming a must. They involve substantial budgets.
The success rate is only 35%. This statistic underlines the extreme complexity. For Class III devices, a simultaneous multi-country approach becomes necessary. You spread the commercial risk. You accelerate penetration despite capital-intensive investment.
Examples of Class III devices: cardiac devices, life-support systems, active implants.
Optimized sequential roadmap
The natural temptation is to go for “France First”. Patriotism, familiarity with the domestic market: these reasons seem logical. Field data reveal a different reality.
Pre-launch phase: 12 months, budget 80,000 euros. Implement ISO 13485, design controls, risk management. This phase structures everything that follows.
First contract – Germany: 18 months, budget 120,000 euros. Why Germany? It’s Europe’s biggest market. Notified bodies are experienced. The average lead time is 13 to 15 months. You have immediate access to 83 million consumers.
Second contract – Italy: 12 months, budget 90,000 euros. Italian organizations are more forgiving. Queues are shorter. You quickly gain market share. Italy offers 60 million additional consumers.
Third market – France: 10 months, budget 75,000 euros. At this stage, you benefit from familiarity with the ANSM. Local reimbursement paths become accessible. You validate your commercial traction before attacking your domestic market.
Total: 52 months and 365,000 euros. This sequence seems long. Let’s compare it with the classic error.
Strategy comparison
The “France Only” approach takes 18 months and costs 450,000 euros. It generates an ROI in year 2 of only 120%. You gain access to 67 million consumers.
The “Germany then France” sequence takes 30 months and 365,000 euros. It delivers a year 2 ROI of 850%. You gain access to 150 million consumers. That’s 7 times more.
This difference is explained by access to a much larger German market. You multiply volumes, validate the business model and then attack France with proven commercial traction.
Does your device class justify a sequential or simultaneous approach? This question deserves an honest answer. It determines your need to raise capital. It structures your capital allocation.
To explore European funding opportunities, discover our analysis of the €553.5 million in e-health startup funding opportunities in Europe.
Notified body negotiations – what no one is saying
Choosing a notified body is not an administrative formality. It’s a strategic decision with major financial implications. The difference between a saturated organization (18 months) and an optimized one (13 months) represents a saving of 100,000 euros. You save 5 months in time-to-market.
Yet 72% of startups neglect this critical step. They submit to the first organization that comes along. This is a costly mistake.
Stage 1: Pre-identification (8 weeks before)
During this phase, check the organization’s technical scope. Is it competent for your class IIa, IIb or III? Then ask for a written capacity statement. This document guarantees a realistic deadline.
Systematically benchmark 3 to 4 competing notified bodies. Compare deadlines and rates. Pre-identification costs between 2,000 and 5,000 euros. It avoids rejection for scope incompatibility. You save around 30,000 euros.
Step 2: Pre-submission meeting (4 weeks before)
During this meeting, you present your design, your classification and your evidence strategy. The notified body then confirms the complete requirement list. This happens BEFORE your official submission.
The statistics speak for themselves. A well-executed pre-submission meeting saves 40% of the total examination time. It reduces rejections by 50%. For an investment of 5,000 to 10,000 euros, you save 50,000 euros. You save 4 to 6 months.
However, according to theEU Notified Bodies Survey 2025, 72% of startups do not carry out a pre-submission. Often through ignorance. Sometimes it’s because we want to save these initial costs.
This false economy costs tens of thousands of euros in overhaul cycles.
Stage 3: Checkpoint documentation (2 weeks before)
This approach involves submitting a draft of your technical file. The notified body returns informal comments. This happens before the file is formally registered.
This tactic drastically reduces rejections for incomplete submission. These rejections account for 50% of cases, according to EU 2025 data. The cost of this checkpoint is generally included in the pre-submission meeting. No additional cost.
Actual costs of notified bodies in 2025
Let’s talk frankly about rates for a Class IIb device. Technical file review is billed at between 250 and 430 euros per hour. The duration varies between 8 and 40 days, depending on complexity. This represents between 16,000 and 138,000 euros.
A quality system audit costs between 1,900 and 8,400 euros. Allow 2 days for on-site auditing. Issuing the certificate represents a fixed cost of between 1,000 and 6,900 euros.
Total costs: between €80,000 and €150,000. These amounts do not include clinical studies, which remain your responsibility.
Negotiations possible on rates
A little-known insight into the field: notified bodies accept discounts of 20 to 30%. The condition? A commitment volume. For example, 3 devices to be certified over 24 months.
Other levers include submission timing. The first quarter is generally less saturated. Standardization of multi-product documentation also enables the organization to gain in efficiency.
Have you ever contacted a notified body to obtain a capacity statement? Or are you waiting to finalize your technical file? This difference in approach separates startups that optimize their trajectory from those that suffer avoidable delays.
Realistic budget and critical milestones – actionable roadmap
“How much will certification cost me?” This is THE question every founder asks during a first pre-levée audit. The answer depends on the class of the system. Above all, it depends on how rigorously you structure your critical milestones.
Budget breakdown Class IIa-IIb (single contract)
Regulatory consulting: 30,000 to 50,000 euros, months 1 to 6, high criticality. This consulting structures your entire approach. It avoids misclassification. It prevents insufficient evidence.
ISO 13485 quality system: 40,000 to 60,000 euros, months 1 to 8, high criticality. Without a compliant quality system, no certification is possible. It’s the foundation for everything else.
Clinical evidence: 80,000 to 150,000 euros, months 6 to 14, high criticality if IIb. This clinical study becomes critical if your device cannot rely solely on existing literature.
Pre-submission meeting + initial NB fees: 10,000 to 15,000 euros, months 8 to 10, criticality. This stage validates your strategy before any major financial commitment is made.
Technical documentation: 50,000 to 80,000 euros, months 8 to 12, high criticality. It forms the core of the dossier submitted. Its quality determines the success of the examination.
Notified body certification costs: 80,000 to 150,000 euros, months 12 to 18, criticality. This is when the official clock starts.
Post-market surveillance: 10,000 to 30,000 euros, month 18+, medium criticality. Mandatory to maintain certification.
Class totals
Class IIa: €250,000 to €350,000 over 15 to 18 months.
Class IIb: 350,000 to 500,000 euros over 18 to 22 months. The discrepancy is essentially due to stricter clinical evidence requirements.
Hidden costs often overlooked
SaMD software compliance: €20,000 to €100,000 depending on the complexity of your system.
Cybersecurity assessment: 15,000 to 40,000 euros if your device is connected.
Translation of technical documents: 5,000 to 15,000 euros per language. Multiply by the number of target markets.
Legal fees: 10,000 to 25,000 euros. Including contracts with the notified body and IP protection.
Buffer contingency 20%: 50,000 to 100,000 euros. All experienced project managers use it to absorb unexpected events.
For a multi-country strategy covering 3 European markets: realistic total budget between €450,000 and €650,000.
ROI modeling Year 2
That may seem like a lot of money. Compare it to the return on investment generated.
Year 1: German certification obtained at t+18 months. Revenue generated: 2.5 million euros thanks to a broad addressable market.
Year 2: French certification acquired at t+30 months. Cumulative revenue: 8.4 million euros thanks to dual geographic presence.
ROI Year 2: 850% versus 120% in a “France Only” scenario. This gap shows that strategic patience outweighs domestic urgency.
5 critical milestones to watch out for
Milestone 1 (Month 6): QMS internal audit completed. Green light to submit to the notified body.
Milestone 2 (Month 10): pre-submission meeting held. Requirement list confirmed.
Milestone 3 (Month 14): technical file submitted. The regulatory clock formally starts.
Milestone 4 (Month 18): certification obtained. Commercial launch authorized.
Milestone 5 (Month 24): post-market data collected. First certification maintenance cycle completed.
Can your current runway absorb an 18-30 month roadmap? Or do you need to raise intermediate funds to secure this trajectory? This financial question determines the optimal sequence between development, certification and marketing.
Regulatory compliance = competitive advantage, not obstacle
Structuring an optimized medical device certification strategy is not a matter of luck. It’s a question of clear milestones and strategic market selection. The 501 MedTech startups analyzed reveal a massive gap. Some suffer from regulation. Others turn it into a competitive advantage.
The figures speak for themselves
The 57% gap between hosted applications and issued certificates confirms that the majority are failing. They get bogged down in the process. Avoiding the 6 documented errors could save between 150,000 and 350,000 euros.
Opting for an optimized geographic sequence Germany-France generates a ROI 7 times higher. You considerably expand the addressable market. You demonstrate your traction before lifting.
The JuliaShift action framework to optimize your certification
First pillar: classification audit via pre-submission meeting. Cost: 5,000 to 8,000 euros. This small investment confirms your regulatory class. It avoids a costly reclassification along the way.
Second pillar: strategic selection of the notified body. Get a confirmed capacity statement. It guarantees a realistic deadline. You avoid an 18-month tail.
Third pillar: sequential roadmap. Focus on Germany for Class IIb devices. Capitalize on the experience of local organizations. Benefit from market size.
Pillar four: lucid budgeting. Anticipate 350,000 to 500,000 euros for a Class IIb device. Watch for critical milestones at months 6, 10, 14, 18 and 24.
Final question for your trajectory
Does your certification roadmap incorporate these 4 optimization levers? Or is it still exposed to the 6 classic budget-busting mistakes?
The answer determines your ability to obtain certification. Above all, it determines your ability to raise funds. It proves to investors that your regulatory path is under control.
Startups that fail don’t lack innovation. They lack operational rigor when it comes to compliance. Successful startups turn compliance into a barrier to entry. Their competitors can no longer catch up.
Free 30-minute pre-emergence audit: diagnose your regulatory blind spots and structure your optimized budget roadmap.
Reserve your niche now. Turn compliance into a competitive advantage rather than a financial hurdle.
🎯 Going further
Are you structuring a MedTech fundraiser?
Download our free strategic reports:
- BPI France 50-point compliance checklist
- Timeline 0-6 months pre-emergence
- 3 startup cases (seed → series A)
- Frameworks valorisation multiples Revenue
📥 Download your free reports → Blueprint MedTech
About the author
Nicolas Schneider is the founder of JuliaShift, a consulting firm specializing in digital healthcare transformation and MedTech regulatory compliance. With 25 years’ experience in the medical sector, including 17 years with the French Army Health Service, Nicolas has supported e-health SMEs in their European expansion. His JuliaShift methodology combines Lean Start-up and regulatory requirements to structure optimized roadmaps without budget explosion.